A new, novel therapy for people suffering with Parkinson’s disease is drug free and requires no anesthetic, no incision, and no hospital stay.
If that sounds too good to be true, then you’ll want to keep reading. Because the results of a published clinical trial on the therapy were so convincing the FDA gave this therapy its stamp of approval.
The therapy I’m talking about is magnetic resonance image-guided focused ultrasound. And here’s what you need to know…
Current treatments for Parkinson’s disease leave a lot to be desired. They include medications and deep brain stimulation (DBS) where electrodes are inserted into the brain. Both can induce unpleasant side effects. For one thing, medications can cause uncontrollable, erratic muscle jerks, hallucinations, and compulsive behaviors. While DBS carries with it a small risk of infection and brain bleeding.
Focused ultrasound has none of these negative reactions.
What Is Focused Ultrasound?
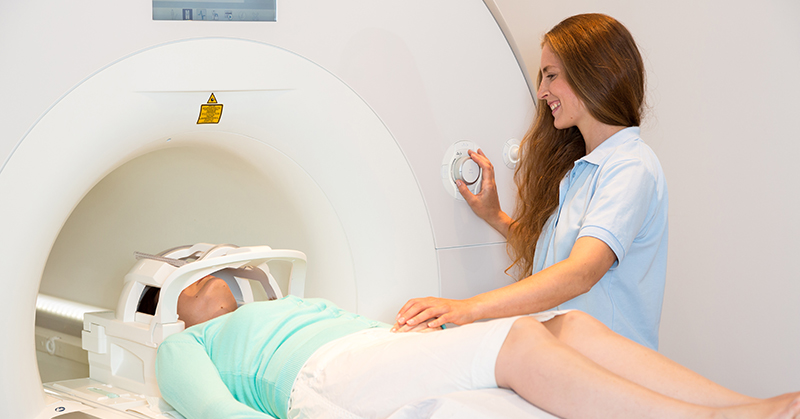
For this treatment the patient lies in an MRI scanner, wearing a special helmet. The helmet transmits ultrasonic energy through the skull into an area that helps control regular voluntary movement. Meanwhile, images from the scanner provide a temperature map of the area being treated to precisely pinpoint the target and to apply a high enough temperature to diseased tissue.
During the procedure, the patient is fully awake and can talk with the medical team. This allows doctors to monitor the immediate effects of the treatment in real time and tweak it as needed.
Often patients experience relief from Parkinson’s tremors, rigidity in the legs and arms, and side effects from medications that cause involuntary, erratic movements called “dyskinesia” before the procedure is even completed.
Researchers led by the University of Maryland School of Medicine (UMSOM) put the procedure through its paces by enrolling 94 patients with moderate Parkinson’s who weren’t responding well to medications.
They randomly assigned the patients to an active (treatment) group or control group. Patients received either one session of focused ultrasound on the brain area that controls the side of their body where symptoms are more severe, or a sham procedure where ultrasound isn’t activated. As well as at UMSOM, patients were treated at fifteen sites across North America, Asia, and Europe.
The results were very encouraging.
Immediate Improvement For 70 Percent
After three months follow up nearly 70 percent of patients in the treatment group had a successful outcome with a significant improvement in tremors, mobility, and other physical symptoms compared to 32 percent in the control group.
The treatment group had an immediate improvement of at least three points on a standard Parkinson’s assessment scale, which is used to measure tremors, walking abilities, and rigidity in the legs and arms – compared to a 0.3-point improvement among controls.
Participants did experience mild side effects such as headaches, dizziness, and nausea, but these quickly resolved. Some had slurred speech, problems walking, and loss of taste as a result of the treatment, but these disappeared within a few weeks.
Perhaps most exciting, two-thirds of those who responded well to the treatment continued to have a successful response one year later. Study author Howard Eisenberg, M.D. explained, saying, “These results are very promising and offer Parkinson’s disease patients a new form of therapy to manage their symptoms. There is no incision involved, which means no risk of a serious infection or brain bleeding.”
The dean of UMSOM, Mark Gladwin, added: “We are on the edge of the frontier with focused ultrasound, as ongoing research evaluates the procedure in different brain areas affected by Parkinson’s. Researchers also are studying how focused ultrasound could be used to temporarily open the blood-brain barrier to help experimental Parkinson’s treatments, like immunotherapy, get into the brain more easily.”
One patient who has already benefited from the procedure is 41-year-old Melanie Carlson.
“One Of The Best Years Of My Life”
Diagnosed with Parkinson’s disease in 2020, Ms. Carlson opted for this novel treatment because medications to control the condition were causing severe side effects.
“Focused ultrasound was really transformative,” she said. “So many of my fine motor skills have returned. I’m putting on eyeliner again and taking showers again without falling. This honestly feels like one of the best years of my life. I just feel so fortunate. I hope more people can benefit from this procedure.”
How To Get Focused Ultrasound
The focused ultrasound device, called Exablate Neuro, was manufactured by Insightec and is available at the University of Maryland Medical Center (UMMC). UMMC is one of a handful of sites in the U.S. with the capabilities and expertise to utilize the therapy. For one thing, the treatment requires a multi-disciplinary team, including a neurosurgeon, movement disorder neurologist, and neuroradiologist.
Currently, focused ultrasound is not covered by insurance, including Medicare. What’s more, the FDA has only approved the therapy to treat one side of the brain, so it may be more appropriate in patients with symptoms predominantly on one side.
However, a clinical trial is underway delivering focused ultrasound in two sessions, six months apart, for both sides of the brain. According to Dr. Eisenberg, “So far, we’ve had promising results.”
I’m not surprised. Focused ultrasound has been used with success against numerous cancers by pioneering integrative medical teams so it’s about time that conventional medicine begins to look at other ways to apply this healing technology. Let’s hope, for the sake of loved ones suffering from Parkinson’s, that the researchers press forward, and the FDA doesn’t delay in approving focused ultrasound if it continues to be effective.
I’ll keep you posted on any new information when it becomes available.
Best Regards,
The Awakening From Alzheimer’s Team